 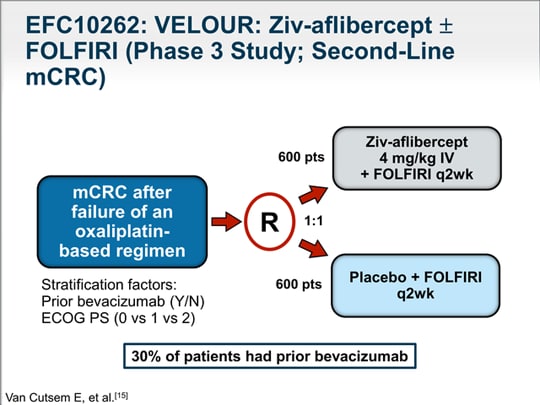
Moreover, the 24-month PFS rate in the investigative arm was 48.3% versus 18.6% in the control arm. The median progression-free survival (PFS) was more than doubled with pembrolizumab versus chemotherapy, at 16.5 months versus 8.2 months, respectively. 0002) compared with investigator’s choice of oxaliplatin, leucovorin, and fluorouracil (FU mFOLFOX6) with or without bevacizumab (Avastin) or cetuximab (Erbitux) or irinotecan, leucovorin, and FU with or without bevacizumab or cetuximab. 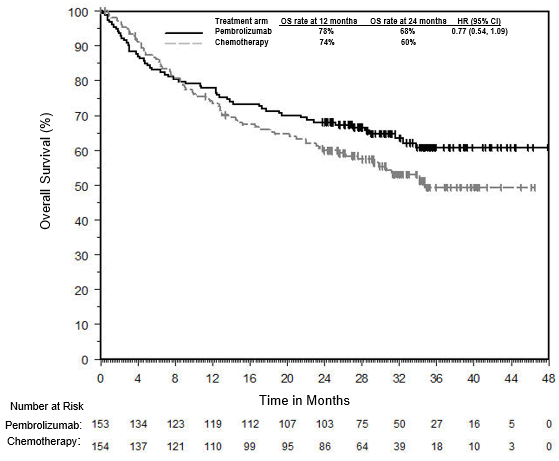
The decision was based on data from the pivotal phase 3 KEYNOTE-177 trial (NCT02563002), which demonstrated that the immunotherapy resulted in a significant 40% reduction in the risk of disease progression or death (HR, 0.60 95% CI, 0.45-0.80 P =. In addition, there was no treatment-related death in the pembrolizumab group.The European Commission has approved single-agent pembrolizumab (Keytruda) for use in the frontline treatment of patients with metastatic microsatellite instability–high (MSI-H) or mismatch repair deficient (dMMR) colorectal cancer (CRC). 
These included diarrhea (2%), fatigue (2%), and anemia (1%) in the pembrolizumab group. Treatment-related adverse events for Grade 3 or worse occurred in 22% of the pembrolizumab group versus 66% in the chemotherapy group. The median overall survival for pembrolizumab (median not reached, 95% confidence interval : 49.2-NR) versus chemotherapy (median 36.7 months, 95% CI: 27.6-NR) did not show statistical difference. The median follow-up period was 44.5 months (interquartile range : 39.7-49.8). The demographic of both study groups was well-balanced. At data-cutoff, 36% of the patients in the chemotherapy group crossed over to receive pembrolizumab in the intention-to-treat group. Patients whose disease progressed while receiving chemotherapy were allowed to cross over to pembrolizumab for up to 35 treatment cycles. A total of 307 patients were enrolled to receive either study treatment (n=153) or chemotherapy (n=154). Patients who have received adjuvant therapy for colorectal cancer 6 months prior to randomization were also included. Patients were eligible if they had previously untreated microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer. In-Depth :This was an open label, randomized, phase III study. Relevant Reading: Pembrolizumab in microsatellite instability-high advanced colorectal cancer Overall, this study demonstrated that although prior reported analysis indicated a large progression-free survival benefit and quality of life benefits, the overall survival was not different likely owing to substantial cross-over and off-label use of immunotherapy.Ĭlick to read the study in The Lancet Oncology The high cross-over rate could have contributed to an improvement in the overall survival rate in the chemotherapy group. 36% of the patients in the chemotherapy group crossed over to receive pembrolizumab and 24% received off-study anti-PD-1/anti-PD-L1 therapies. It is important to note that the crossover design could have contributed to the absence of significant overall survival benefit for pembrolizumab. Diarrhea, fatigue, and anemia were common grade 3 or higher treatment-related adverse events that were reported. At final analysis, the median overall survival as per the intention-to-treat analysis was not statistically different. This study presents the final overall survival analysis of the KEYNOTE-177 study. Based on the initial data from the KEYNOTE-177 study, pembrolizumab showed improved progression-free survival compared to chemotherapy in patients with previously untreated microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer. Study Rundown:The presence of mismatch repair deficiency in colorectal cancer can help inform the surgical and systemic treatment for patients.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
